
Endoscopic interventions in the management of the gastroesophageal reflux: a narrative review
Introduction
Gastroesophageal reflux disease (GERD) is one of the most common gastrointestinal (GI) ailments in the United States. It is considered to be predominantly an anatomical problem. The thickened circular muscular layer of the distal esophagus comprises the lower esophageal sphincter (LES)—a 2.5 to 4.5 cm in length structure, spanning the diaphragm, by which is attached via the phrenoesophageal membrane (1-4). The LES, together with the diaphragmatic crura and the angle of His, forms a flap valve which contributes to the barrier function of the gastroesophageal junction (GEJ). Additionally, esophageal peristalsis facilitates the clearance of gastric refluxate from the esophagus, overcoming the barrier mechanisms.
The prevalence of GERD has been estimated to be between 18.1–27.8% in North America, with evidence showing a steady increase in disease prevalence (5). It is estimated that this condition affects at least 60 million Americans with symptomatic GERD, leading to significant decrease is quality of life and loss of productivity (6). The chronic exposure of esophageal mucosa to gastric refluxate leads to numerous complications, including esophagitis, strictures, Barrett’s esophagus and potentially esophageal cancer (7). Initial treatment of GERD includes diet and life style modification, as well as medical therapy, predominantly involving acid suppression agents. None of these interventions address anatomical alterations of the GEJ and rather focus on acid suppression. In reality, this simply masks the problem. Up to 40–50% of patients do not have satisfactory reflux control with conservative therapy. However, surgical correction is reserved for severe or refractory cases. We present the following article in accordance with the Narrative Review reporting checklist (available at https://aoe.amegroups.com/article/view/10.21037/aoe-21-52/rc).
Objectives
With the magnitude of the problem, only a miniscule number of fundoplications are performed annually in the United States—about 24,540 in 2013 (0.04% of the affected population) (8). At our current operative rate, this problem would take over 2000 years to fully address. The low number of fundoplications performed annually is likely due to negative connotations on perceived invasiveness of the procedure, reserving surgical referrals as a last resort measure after medical therapy is fully exhausted. This huge disparity between the patient population and surgical volume creates a substantial treatment gap and offers an opportunity for less invasive, endoscopic interventions to address the anatomic basis of the problem and improve outcomes (9,10). Here, we thought to review currently in practice endoscopic interventions aimed at correction of the anatomical basis of GERD.
Methods
The PubMed electronic database was utilized and articles March 1999 and March 2021 were searched. Our search strategy involved terms specific to the disease process (i.e., GERD, reflux) the management of said disease (i.e., endoscopic fundoplication, endoscopic interventions for reflux), and specific endoscopic modalities [i.e., transoral incisionless fundoplication (TIF), Medigus ultrasonic surgical endostapler (MUSE), Stretta]. Additionally, studies referenced in our acquired articles from the database were individually searched for additional references.
Our inclusion criteria were fairly broad, and contained: (I) randomized control trials; (II) meta-analyses; (III) non-randomized prospective studies; (IV) retrospective studies. Our study focused on adult populations, and exclusion criteria were articles including patients below the age of 19. In total, we included three systematic reviews, four meta-analyses, and four randomized control trials and other publications.
Endoscopic procedures for management of GERD
TIF
TIF is one of the best established endoscopic anti-reflux interventions currently in practice with over 30 thousand procedures performed to date (see Figure 1 and Video 1 demonstrating a TIF procedure on 01/30/2020). The initial generation of this procedure, the transoral-endoluminal fundoplication (ELF), was introduced in the mid-2000s with the EsophyX device (EndoGastric Solutions, Redmond, Washington, USA), and is the earliest form of TIF, involving gastric fundus-to-fundus fixation (11). In the original form, ELF involved gastric to gastric fasteners below the Z-line, and did not include a wrap. Gastric-gastric plication was utilized in this initial model as the investigators were concerned with the potential injury to the distal esophagus. Outcome studies soon revealed a high rate of recurrence with ELF, requiring traditional operative fundoplication for revision (12,13). Several years later, ELF evolved into TIF 1.0, involving esophagus-to-gastric fundoplication with fasteners placed 1 cm above the Z line, while still avoiding a formal wrap. At this stage, the number of fasteners deployed was found to be associated with a stronger anti-GERD response. Sixty percent of those who underwent TIF with a larger number of fasteners deployed (mean 12 fasteners) were found to remain of proton-pump inhibitors (PPI) at 6 months (P=0.018) compared to those with less fasteners deployed (mean 9 fasteners), as seen by Testoni et al. (14). TIF 1.0 was then superseded by TIF 2.0 utilizing the EsophyX device, which accomplished a 270-degree gastric wrap around the esophagus, utilizing at least 20 fasteners. The next generation model, the EsophyX-Z device, was developed with automatic deployment of fasteners. The most recent development, the EsophyX-Z+, has compatibility with a greater array of endoscopes.
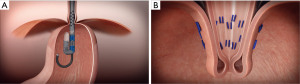
In 2018, the randomized prospective TEMPO trial analyzed outcomes of 63 participants, and demonstrated that greater than two-thirds of patients remained off daily PPIs 5 years after having undergone the TIF 2.0 procedure (15). The elimination of troublesome regurgitation was found to be 86% at 5-year follow-up in this study, based on the reflux disease questionnaire. The TEMPO trial also found that within 5 years, only 5% of those who underwent TIF 2.0 required revision with surgical fundoplication. Furthermore, TIF did not negatively affect the outcomes of surgical revision—indicating that TIF could be used as a bridging therapy without unfavorable consequences. Interestingly, the trial also demonstrated the economic advantage of the TIF procedure. The total cost for a laparoscopic Nissen fundoplication (LNF) was nearly twice as much—$124,000, compared to $66,000 for TIF during the first 2 years post-operatively. Contrary to this data, a meta-analysis by Huang et al., involving 13 prospective studies, including 616 patients who underwent TIF, demonstrated that PPI usage eventually resumed at reduced dosages for approximately 80% of the patients by 48 months follow-up, suggesting decreased efficacy over time (16). This compares to 11% of patients resuming PPI therapy at 48-month follow-up after laparoscopic fundoplication in one study (17).
TIF 2.0 has also been demonstrated as an acceptable option for the reduction of small hiatal hernias. Several studies have demonstrated superior outcomes and experiences with TIF for hiatal hernias smaller than 2–3 cm in greatest transverse diameter (18-20). Current standard practice allows TIF 2.0 for the reduction of hernias less than 2 cm in the greatest transverse diameter. Typically, hiatal hernias greater than 2–3 cm are better served with formal surgical (laparoscopic or robotic) hiatal hernia repair (HHR).
In a recent study by Janu et al., the efficacy of laparoscopic HHR combined with TIF 2.0 was assessed in 99 patients (21). The study found improved symptom and quality of life scores at 12-month follow-up. Surveys such as the GERD-Health-Related Quality of Life Questionnaire (GERD-HRQL) were utilized and demonstrated median score improvements of 19 points at 6 months, essentially meaning minimal to absent GERD symptoms. Seventy-four percent of their patients were not using PPIs at the end of follow-up, comparable to TIF 2.0 alone, but still inferior to traditional laparoscopic fundoplication. The procedure was also not associated with dysphagia or gas bloat syndrome, common in laparoscopic fundoplication (22). Notably, it has been demonstrated that postoperative dysphagia rates after laparoscopic fundoplication are very similar to rates in those on PPI therapy alone. In the recent meta-analysis by McKinley et al., reviewing 5 randomized controlled trials (RCTs) involving 1,228 patients, there was no statistically significant difference in the rates of dysphagia in the laparoscopic fundoplication group versus PPI group at 5 years, with a relative risk of 0.92 (23).
Whereas the short and medium-term outcomes for TIF 2.0 with the EsophyX device appear acceptable, long-term data is insufficient. This prompted the European Society of Gastrointestinal Endoscopy (ESGE) to release guidelines voicing caution to the use of TIF as a global alternative to PPI therapy or anti-reflux surgery in 2020 (24). The ESGE guidelines state that there may be a place for TIF in those with mild GERD, who are unwilling to remain compliant on PPIs or undergo surgical intervention. Whereas the rates of post-procedural dysphagia are similar between patients after fundoplication or on PPI therapy alone, gas-bloat syndrome, a common post-operative issue after laparoscopic fundoplication seen in up to 19% of patients following laparoscopic fundoplication, seems to not be observed after HHR combined with TIF, or after TIF alone (25). Long-term efficacy of TIF 2.0 seem to decrease with time, suggested by the persistence of PPI. New long-term studies, factoring in the lifelong cost of PPI use in these patients during disease relapse, are needed to fully determine the overall cost effectiveness of the procedures when comparing TIF and laparoscopic fundoplication. Importantly, TIF was shown not to interfere with the ability to perform laparoscopic fundoplication down the line and as such can be utilized as a bridging therapy.
Additionally, with the rise in popularity of peroral endoscopic myotomy (POEM) as the primary mode of treatment for esophageal achalasia, there is a unique group of patients with post-POEM GERD. Whereas the majority of these subjects are successfully managed with medical therapy, similar to patients with traditional GERD, for those with failure of the treatment, surgical intervention is contemplated. TIF offers a unique opportunity for reflux control, keeping the patient in the realm of endoscopic therapy without crossover to surgery. In one small study, evaluating 5 patients with post-POEM GERD, who underwent TIF (26). Tyberg et al. found that at 27-month follow-up, 100% of patients discontinued PPI usage.
MUSE
MUSE is another new device for TIF. While the EsophyX device creates a 250–300-degree omega-shaped fundoplication around the GEJ, the MUSE device is an approximate 180-degree anterior wrap, similar to the Dor fundoplication (27). The MUSE device utilizes an endostapler with a cartridge containing five 4.8-mm titanium staples, endoscopic ultrasound, two anvil screw funnels, and a ratchet controlled articulating tip. The steps of the procedure include advancing the device into the stomach, retro-flexing into the fundus, retracting the system to 3 cm proximal to the gastro-esophageal junction, clamping the fundal-esophageal tissue, and firing the stapler. It is important to maintain an appropriate tissue thickness using the endoscopic ultrasound before firing the stapler.
In terms of durability and morphologic strength, the preliminary literature of MUSE fundoplication was limited to animal and ex-vivo studies. An early study using porcine models, found that the endoscopic anterior fundoplication was intact in 12 pigs during post-sacrifice autopsy who underwent the procedure at 2, 4 and 6 weeks prior (28). An ex-vivo study, performing MUSE fundoplication on porcine esophagus and stomach, demonstrated good reflux control with significantly higher intragastric pressures required to detect stomach contents (methylene blue dyed normal saline), in the esophagus (29). Compared to the EsophyX device, the literature on MUSE outcomes is still sparse—there are very few small studies that exist to date. However, these few studies demonstrate promising short-term outcomes of MUSE fundoplication (30). Testoni et al. found a 65% cessation of PPI after 12 months following MUSE fundoplication, as well as a 70% reduction in GERD-HRQL scores. A 5-year follow-up study looking at 13 patients who underwent MUSE fundoplication found that 54% had eliminated use of PPIs, while 23% reduced PPI use by at least 50% (31). In terms of comparative studies to EsophyX, there is only one meta-analysis comparing a few clinical end-points between the two procedures. Due to limited data, only two MUSE studies were available for comparison with thirty EsophyX studies. Such, McCarty et al. found the mean reduction of the number of reflux episodes post-intervention to be 3.61 and 3.97 for TIF 2.0 and MUSE, respectively (32). Similarly to TIF, the 2020 ESGE guidelines have also cautioned against the routine use of MUSE in clinical practice due to the paucity of data on safety and effectiveness, and suggested its use in clinical trials only (24).
The Stretta system
The Stretta system was developed by Mederi Therapeutics in the early 2000s. It is an endoscopic modality for GERD that involves radiofrequency energy application to the LES and gastric cardia, theoretically causing tissue fibrosis and neural ablation, affecting, in turn, tissue compliance (33). Arts et al. analyzed lower esophageal pressures and measured compliance after Stretta and sham procedure in 22 patients. They found a significant decrease in mean compliance in the Stretta group, from 17.8 mL/mmHg pre-procedure to 7.4 mL/mmHg post-procedure. In comparison, the compliance of the sham procedure group was approximately unchanged, from 14.0 pre-procedure to 13.3 post-procedure. There is no consensus on the efficacy of Stretta for symptom control in GERD patients. A recent meta-analysis by Fass et al. analyzing 2,468 patients across 28 studies, demonstrated Stretta to decrease the pooled estimate of HRQL scores by a mean of −14.60, as well as reducing the incidence of erosive esophagitis by 24%, and decreasing the pooled estimate of esophageal acid exposure by −3.01 via pH-impedance monitoring (34). However, there was no difference in resting LES pressures. Another meta-analysis by Lipka et al., which analyzed 153 patients across four trials, suggested that the studies’ clinical end-points were limited (35). The authors pointed to inconsistent studies’ design, and small sample sizes. In the pooled analysis from 3 RCTs involving 118 patients, no statistically significant difference in cessation of PPI use after Stretta over sham therapy were evident. Furthermore, in this meta-analysis, GERD-HRQL scores from 2 RCTs, involving 88 patients, did not show a statistically significant difference between the Stretta and sham groups (35). This suggests limited efficacy of the procedure and justifies further long-term analysis for critical appraisal of the results. Complications from the Stretta procedure are minimal, and rarely include transient gastroparesis and reversible erosive esophagitis per literature (36).
Funk et al. analyzed several economic parameters, and found that Stretta seems to be more cost effective than LNF (37). In the authors’ analysis, an incremental cost-effectiveness ratio (ICER) was defined as the ratio between cost and quality-adjusted life year (QALY). In this study, out of all endoscopic and surgical procedures, the ICER was lowest for Stretta at $642.63, $716.84 for TIF, and highest for LNF at $1,067.21. The criticism for Funk’s study, however, is wide application of extrapolations, using medium term follow up data to project 30-year relapse probabilities (37). Currently, Stretta is still a relatively new technology with uncertain clinical applicability. More robust outcomes data is necessary to better assess the position of this modality in the clinical practice. Due to low quality evidence, the 2020 ESGE guidelines suggest Stretta in very select patients, such as those without esophagitis or hiatal hernias (24).
EndoCinch device
Approved in the early 2000s, the EndoCinch suturing system (Bard Endoscopic Technologies, Billerica, MA, USA) was introduced as an endoscopic option for anti-reflux management. The EndoCinch system involves lowering the device to the level of the GEJ, and applying negative pressure via vacuum suction, thereby pulling tissue into the chamber. Two sets of sutures are then fired over the tented tissue, creating a full-thickness plication. Several early studies have demonstrated improved GERD symptoms and reduced PPI use in the short term (38-40). Nevertheless, long-term data demonstrated poor results, likely related to poor plication durability. Schiefke et al. followed 70 patients who underwent the procedure, and found that 80% of patients were considered treatment failures at 18-month (41). This was attributed to loss of plicating suture, as only 17% of patients on surveillance endoscopy retained all sutures, while 26% of patients demonstrated no remaining sutures whatsoever. Similarly, high rates of suture loss were seen in a prospective trial by Domagk’s group, demonstrating 19.2% total suture loss at 6-month follow-up in a group of 26 patients (42). Likewise, Abou-Rebyeh et al. found in 38 patients, 90% of sutures were lost at 1-year follow-up (43).
The safety-profile of EndoCinch seems to have been acceptable, with low rates of adverse events. A systematic review by Chen et al. analyzing 629 patients across 12 studies, highlighted pharyngitis to be the most common complication of the procedure (44). The review also found post-operative bleeding rates of 2–11%, and one case of mucosal tear. Despite the low complication rate, the EndoCinch has fallen out of favor due to high treatment failure and is no longer in clinical practice. This technology serves primarily as a historical example of one of the earlier endoscopic plicating devices for the management of GERD.
Enteryx device
Enteryx procedure (Boston Scientific, Marlborough, MA, USA) is an endoscopic modality for GERD, no longer available on the market. It involved the injection of 8% ethyl vinyl alcohol dissolved in dimethyl sulfoxide into the LES (45). The liquid injected would coalesce and help aid in forming an anti-reflux barrier. The procedure was recalled in 2005 secondary to 11 complications related to esophageal perforation, and one death secondary to aortic injury (46). There have been several other modalities involving endoscopic injection of bulking agents, none of which have been FDA approved for human use for GERD treatment.
Gatekeeper Reflux Repair System
The Gatekeeper Reflux Repair System (Medtronic, Inc., Shoreview, MN, USA), is an endoscopic injection technique for the management of GERD that is no longer available in clinical practice. It utilized a substance known as polyacrylonitrile-based hydrogel that is injected into the esophageal submucosa at the LES (47). In a prospective trial by Fockens et al., 143 patients were enrolled and studied. Twenty-five lead-in patients, 75 randomized patients who underwent the Gatekeeper Reflux procedure, and 48 sham patients were analyzed. A lead-in was defined as a non-randomized patient who was chosen by the investigator to undergo the Gatekeeper Reflux procedure. Lead-in patients were not included in efficacy analysis. Heartburn symptoms based on HRQL scores were significantly improved at 6-month follow-up in the Gatekeeper group, however, the sham group had similarly improved HRQL scores at 6 months (HRQL 13.7 and 16.4, respectively, P=0.146). Out of those who underwent the Gatekeeper Reflux procedure, two had esophageal perforations. The study highlighted the lack of efficacy of the Gatekeeper procedure compared to sham.
OverStitch device
The OverStitch (Apollo Endosurgery, Austin, TX, USA) is a form of endoscopic suturing device, utilizing traditional surgical technique that was approved by the FDA in the early 2010s (Figure 2). It is primarily used for closure of mucosal or full thickness GI defects in various procedures, stent fixation and fistula closures (48,49). It has also shown some potential utility in treating reflux after esophagectomy. Yanagimoto and colleagues utilized the OverStitch device to create an anti-reflux valve in four pigs who underwent esophagectomy with gastric conduit reconstruction. The authors concluded the procedure to be successful based on morphologic and functional assessments by analyzing a measured reflux angle (50). One study in 2018 demonstrated the use of OverStitch in four patients with post-esophagectomy reflux (51). The study outcomes were limited, but subjective symptoms were reportedly improved, with decreased DeMeester scores. While more extensive studies are necessary to validate this new utility for the OverStitch device, it showcases the ongoing development of new technologies and uses.

Anti-Reflux Mucosectomy (ARMS) procedure
ARMS was originally described by Satodate and colleagues in 2003 (52). A circumferential mucosal resection of the distal esophagus for high-grade dysplasia was performed on a patient with Barrett’s esophagus and GERD. The procedure would remove the high-grade dysplasia, while theoretically creating a stricture of the distal esophagus secondary to scar formation (53). This would thereby potentially lessen the symptoms of reflux. The procedure was ultimately successful, resulting in resolution of Barrett’s and GERD. In the current application ARMS procedure involves partial circumference endoscopic resection, either by endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) of the mucosa at the GEJ termed crescentic ARMS (ARMS-C), essentially creating a stricture. A recent study by Yoo et al., involving 33 patients who underwent ARMS-C, found that 63% of patients discontinued using PPIs, while 30% of patients reduced their PPI dose at 6-month follow-up (54). Other than two patients who underwent balloon dilatation secondary to stricture formation, there were no other complications. ARMS may be uniquely suited for patients with loss of fundus due to previous surgical intervention, like patients after gastric bypass or sleeve gastrectomy. One case series by Debourdeau et al. shares results of ARMS in 6 patients who underwent laparoscopic sleeve gastrectomy (55). The authors experienced a response of >50% in the reduction of GERD-HRQL scores at 3 months, in 5 of the 6 patients (83%). Citing technical challenges in re-do ARMS procedure, Inoue and colleagues developed a new technique, termed anti-reflux mucosal ablation (ARMA) (56). Ablation of the mucosa is performed utilizing a triangle-tip knife in a spray coagulation mode. Twelve patients underwent ARMA, and the mean GERD-HRQL score improved from 30.5 prior to ARMA, to 12 at 2 months post-procedure.
POEM and fundoplication (POEM + F)
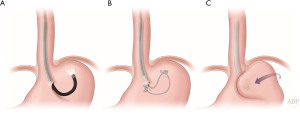
A new intriguing paper was recently made available by Inoue group. During the POEM procedure, the authors advanced the endoscope into the peritoneal cavity (57), fixating gastric fundus onto the GEJ, creating a partial fundoplication (Figure 3). Although it was only a technical report of 21 patients, without assessment of the degree of reflux, 2 months after the procedure twenty (95.2%) patients maintained the wrap. Whereas the current procedure has a very narrow applicability as an adjunct intervention to POEM, it may be laying the pathway for the future development in the area, crossing the boundaries of purely endo- or extraluminal interventions into the natural orifice transluminal endoscopic surgery (NOTES) realm.
Conclusions
With a wide array of endoscopic modalities to choose from, one should be familiarized with the pros and cons of each technology. The characteristics of each intervention are summarized in Table 1. Of the discussed procedures, TIF is the most extensively studied. TIF 2.0 with the EsophyX device has shown a significant improvement in reflux symptoms, and has demonstrated substantial elimination of PPI use in patients’ post-procedure. However, TIF is not indicated for hiatal hernias >2 cm and has higher rates of reflux compared to laparoscopic fundoplication. MUSE demonstrates similar improvement in GERD symptoms/HRQL scores; however, the data is substantially sparse compared with TIF procedure. Consequently, ESGE guidelines have recommended TIF 2.0 for those with mild GERD symptoms, however they recommended against the use of MUSE outside of clinical trials. The limited data on the Stretta device has not shown significant reduction of GERD symptoms or reductions in PPI usage. The EndoCinch, Enteryx system and the Gatekeeper Reflux Repair System are no longer available in the market due to inefficacy and/or safety concerns, and serve as historical examples of endoscopic therapies that may pave the way for future inventions. Although there is minimal data for this usage of the device, the Overstitch has been utilized for those with post-esophagectomy reflux, and has shown some promising results. The ARMS procedure utilizes partial circumferential EMR to form a stricture, which has shown to improve GERD symptoms by theoretically strengthening the anti-reflux barrier. It also has an additional benefit of removing the high-grade dysplasia in Barrett’s esophagus patients. Finally, the POEM + F offers the ability to treat post-POEM reflux in those with achalasia, and is a fascinating new advancement in endoscopic surgery.
Table 1
| Device | Symptomatic relief/PPI usage | Benefits | Shortcomings |
|---|---|---|---|
| TIF | Up to 86 % elimination of reflux symptoms at 5 years | No association with dysphagia or gas-bloat syndrome | Not indicated for hiatal hernias >2 cm |
| Eliminates use of PPIs in greater than two-thirds of patients | Economic advantage in the short-run | Higher rates of reflux recurrence compared to LNF | |
| MUSE | 70% reduction in GERD-HRQL scores | Unclear benefits due to paucity of data | ESGE guidelines recommend against use outside of clinical trials |
| Eliminates use of PPIs in 54% of patients | |||
| Stretta | Lack of statistically significant difference in cessation of PPIs between Stretta and sham group | Unclear given lack of data | Limited studies |
| ESGE recommends very limited use in highly selective patient populations | |||
| EndoCinch | Majority of patients considered treatment failures | No benefit | Poor durability secondary to suture loss |
| Fallen out of favor | |||
| Enteryx | Recalled in 2005. No reliable clinical assessment of GERD symptoms | No clear benefit | High rates of esophageal perforation |
| Gatekeeper Reflux Repair System | Statistically insignificant differences in GERD-HRQL scores between Gatekeeper and sham group | No clear benefit | Cases of esophageal perforation |
| No longer offered by manufacturer | |||
| Overstitch | Subjective improvement in reflux symptoms | Potential utility in those with post-esophagectomy reflux | Few studies with limited outcomes |
| ARMS | 63% of patients discontinued PPI usage at 6-month follow-up | Potential utility in those with high-grade dysplasia and GERD | Symptomatic stricture formation |
| POEM + F | No assessment in reflux symptoms | Potential utility in those with achalasia and GERD | Extremely limited data lacking assessment of reflux symptoms |
ARMS, Anti-Reflux Mucosectomy; ESGE, European Society of Gastrointestinal Endoscopy; GERD, gastroesophageal reflux disease; HRQL, Health-Related Quality of Life; LNF, laparoscopic Nissen fundoplication; MUSE, Medigus ultrasonic surgical endostapler; POEM + F, peroral endoscopic myotomy and fundoplication; PPIs, proton-pump inhibitors; TIF, transoral incisionless fundoplication.
The one limitation of the current data is the lack of objective quantitative data, and heavy reliance on subjective measures such as HRQL scores and patient reported PPI usage post-procedure. Although more objective measures such as pH monitoring, post-procedural impedance studies, are more cumbersome, invasive, and logistically unrealistic for patients to undergo. It is difficult to justify obtaining these studies post-procedurally, especially when patients symptoms have improved, especially from an insurance approval standpoint. As a result, the surgeon has limited indicators to justify using one of these technologies. Encouraging data in multiple studies, demonstrating symptomatic improvement in GERD should serve as a reasonable guide for the foregut specialist in applying this procedure in own practice.
With the increasing prevalence of GERD, the development of new minimally invasive interventions for the management of GERD is of paramount significance. Although endoscopic interventions appear to be in its infancy, there is huge potential, and surgeons should stay abreast of the technology to offer cutting edge treatment to this vast patient population. As for the novel procedures with limited reports, one must consider the clinical scenario, and balance this with the relative lack of alternatives to accumulate data to help this challenging patient population.
Acknowledgments
Funding: This research was funded in part through the NIH/NCI Cancer Center Support Grant P30 CA006927.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Esophagus for the series “New Technologies in Esophageal Surgery and Endoscopy”. The article has undergone external peer review.
Reporting Checklist: The authors have completed the Narrative Review reporting checklist. Available at https://aoe.amegroups.com/article/view/10.21037/aoe-21-52/rc
Peer Review File: Available at https://dx.doi.org/10.21037/aoe-21-52
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aoe.amegroups.com/article/view/10.21037/aoe-21-52/coif). The series “New Technologies in Esophageal Surgery and Endoscopy” was commissioned by the editorial office without any funding or sponsorship. AA served as an unpaid Guest Editor of the series and serves as an unpaid editorial board member of Annals of Esophagus from May 2020 to April 2022. RP served as an unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement:
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Petrov RV, Su S, Bakhos CT, et al. Surgical Anatomy of Paraesophageal Hernias. Thorac Surg Clin 2019;29:359-68. [Crossref] [PubMed]
- Bakhos CT, Patel SP, Petrov RV, et al. Management of Paraesophageal Hernia in the Morbidly Obese Patient. Thorac Surg Clin 2019;29:379-86. [Crossref] [PubMed]
- Hershcovici T, Mashimo H, Fass R. The lower esophageal sphincter. Neurogastroenterol Motil 2011;23:819-30. [Crossref] [PubMed]
- Apaydin N, Uz A, Elhan A, et al. Does an anatomical sphincter exist in the distal esophagus? Surg Radiol Anat 2008;30:11-6. [Crossref] [PubMed]
- El-Serag HB, Sweet S, Winchester CC, et al. Update on the epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut 2014;63:871-80. [Crossref] [PubMed]
- Liker H, Hungin P, Wiklund I. Managing gastroesophageal reflux disease in primary care: the patient perspective. J Am Board Fam Pract 2005;18:393-400. [Crossref] [PubMed]
- Lagergren J, Bergström R, Lindgren A, et al. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med 1999;340:825-31. [Crossref] [PubMed]
- Schlottmann F, Strassle PD, Patti MG. Antireflux Surgery in the USA: Influence of Surgical Volume on Perioperative Outcomes and Costs-Time for Centralization? World J Surg 2018;42:2183-9. [Crossref] [PubMed]
- Triadafilopoulos G, Azagury D. How can we deal with the GERD treatment gap? Ann N Y Acad Sci 2016;1381:14-20. [Crossref] [PubMed]
- Huynh P, Konda V, Sanguansataya S, et al. Mind the Gap: Current Treatment Alternatives for GERD Patients Failing Medical Treatment and Not Ready for a Fundoplication. Surg Laparosc Endosc Percutan Tech 2020;31:264-76. [Crossref] [PubMed]
- Bazerbachi F, Krishnan K, Abu Dayyeh BK. Endoscopic GERD therapy: a primer for the transoral incisionless fundoplication procedure. Gastrointest Endosc 2019;90:370-83. [Crossref] [PubMed]
- Repici A, Fumagalli U, Malesci A, et al. Endoluminal fundoplication (ELF) for GERD using EsophyX: a 12-month follow-up in a single-center experience. J Gastrointest Surg 2010;14:1-6. [Crossref] [PubMed]
- Fumagalli Romario U, Barbera R, Repici A, et al. Nissen fundoplication after failure of endoluminal fundoplication: short-term results. J Gastrointest Surg 2011;15:439-43. [Crossref] [PubMed]
- Testoni PA, Vailati C, Testoni S, et al. Transoral incisionless fundoplication (TIF 2.0) with EsophyX for gastroesophageal reflux disease: long-term results and findings affecting outcome. Surg Endosc 2012;26:1425-35. [Crossref] [PubMed]
- Trad KS, Barnes WE, Prevou ER, et al. The TEMPO Trial at 5 Years: Transoral Fundoplication (TIF 2.0) Is Safe, Durable, and Cost-effective. Surg Innov 2018;25:149-57. [Crossref] [PubMed]
- Huang X, Chen S, Zhao H, et al. Efficacy of transoral incisionless fundoplication (TIF) for the treatment of GERD: a systematic review with meta-analysis. Surg Endosc 2017;31:1032-44. [Crossref] [PubMed]
- Schwameis K, Oh D, Green KM, et al. Clinical outcome after laparoscopic Nissen fundoplication in patients with GERD and PPI refractory heartburn. Dis Esophagus 2020;33:doz099. [Crossref] [PubMed]
- Ihde GM 2nd, Pena C, Scitern C, et al. pH Scores in Hiatal Repair with Transoral Incisionless Fundoplication. JSLS 2019;23:e2018.00087.
- Bell RC, Cadière GB. Transoral rotational esophagogastric fundoplication: technical, anatomical, and safety considerations. Surg Endosc 2011;25:2387-99. [Crossref] [PubMed]
- Ihde GM, Besancon K, Deljkich E. Short-term safety and symptomatic outcomes of transoral incisionless fundoplication with or without hiatal hernia repair in patients with chronic gastroesophageal reflux disease. Am J Surg 2011;202:740-6; discussion 746-7. [Crossref] [PubMed]
- Janu P, Shughoury AB, Venkat K, et al. Laparoscopic Hiatal Hernia Repair Followed by Transoral Incisionless Fundoplication With EsophyX Device (HH + TIF): Efficacy and Safety in Two Community Hospitals. Surg Innov 2019;26:675-86. [Crossref] [PubMed]
- Bakhos CT, Petrov RV, Parkman HP, et al. Role and safety of fundoplication in esophageal disease and dysmotility syndromes. J Thorac Dis 2019;11:S1610-7. [Crossref] [PubMed]
- McKinley SK, Dirks RC, Walsh D, et al. Surgical treatment of GERD: systematic review and meta-analysis. Surg Endosc 2021;35:4095-123. [Crossref] [PubMed]
- Weusten BLAM, Barret M, Bredenoord AJ, et al. Endoscopic management of gastrointestinal motility disorders - part 2: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2020;52:600-14. [Crossref] [PubMed]
- Bakhos CT, Abbas AE, Petrov RV. Tailoring Endoscopic and Surgical Treatments for Gastroesophageal Reflux Disease. Gastroenterol Clin North Am 2020;49:467-80. [Crossref] [PubMed]
- Tyberg A, Choi A, Gaidhane M, et al. Erratum: Transoral Incisionless fundoplication for reflux after peroral endoscopic myotomy: a crucial addition to our arsenal. Endosc Int Open 2018;6:C2. [Crossref] [PubMed]
- Testoni PA, Mazzoleni G, Testoni SG. Transoral incisionless fundoplication for gastro-esophageal reflux disease: Techniques and outcomes. World J Gastrointest Pharmacol Ther 2016;7:179-89. [Crossref] [PubMed]
- Kauer WK, Roy-Shapira A, Watson D, et al. Preclinical trial of a modified gastroscope that performs a true anterior fundoplication for the endoluminal treatment of gastroesophageal reflux disease. Surg Endosc 2009;23:2728-31. [Crossref] [PubMed]
- Gweon TG, Matthes K. Prospective, Randomized Ex Vivo Trial to Assess the Ideal Stapling Site for Endoscopic Fundoplication with Medigus Ultrasonic Surgical Endostapler. Gastroenterol Res Pract 2016;2016:3161738. [Crossref] [PubMed]
- Testoni PA, Testoni S, Mazzoleni G, et al. Transoral incisionless fundoplication with an ultrasonic surgical endostapler for the treatment of gastroesophageal reflux disease: 12-month outcomes. Endoscopy 2020;52:469-73. [Crossref] [PubMed]
- Roy-Shapira A, Bapaye A, Date S, et al. Trans-oral anterior fundoplication: 5-year follow-up of pilot study. Surg Endosc 2015;29:3717-21. [Crossref] [PubMed]
- McCarty TR, Itidiare M, Njei B, et al. Efficacy of transoral incisionless fundoplication for refractory gastroesophageal reflux disease: a systematic review and meta-analysis. Endoscopy 2018;50:708-25. [Crossref] [PubMed]
- Arts J, Bisschops R, Blondeau K, et al. A double-blind sham-controlled study of the effect of radiofrequency energy on symptoms and distensibility of the gastro-esophageal junction in GERD. Am J Gastroenterol 2012;107:222-30. [Crossref] [PubMed]
- Fass R, Cahn F, Scotti DJ, et al. Systematic review and meta-analysis of controlled and prospective cohort efficacy studies of endoscopic radiofrequency for treatment of gastroesophageal reflux disease. Surg Endosc 2017;31:4865-82. [Crossref] [PubMed]
- Lipka S, Kumar A, Richter JE. No evidence for efficacy of radiofrequency ablation for treatment of gastroesophageal reflux disease: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2015;13:1058-67.e1. [Crossref] [PubMed]
- Perry KA, Banerjee A, Melvin WS. Radiofrequency energy delivery to the lower esophageal sphincter reduces esophageal acid exposure and improves GERD symptoms: a systematic review and meta-analysis. Surg Laparosc Endosc Percutan Tech 2012;22:283-8. [Crossref] [PubMed]
- Funk LM, Zhang JY, Drosdeck JM, et al. Long-term cost-effectiveness of medical, endoscopic and surgical management of gastroesophageal reflux disease. Surgery 2015;157:126-36. [Crossref] [PubMed]
- Schwartz MP, Wellink H, Gooszen HG, et al. Endoscopic gastroplication for the treatment of gastro-oesophageal reflux disease: a randomised, sham-controlled trial. Gut 2007;56:20-8. [Crossref] [PubMed]
- Mahmood Z, McMahon BP, Arfin Q, et al. Endocinch therapy for gastro-oesophageal reflux disease: a one year prospective follow up. Gut 2003;52:34-9. [Crossref] [PubMed]
- Filipi CJ, Lehman GA, Rothstein RI, et al. Transoral, flexible endoscopic suturing for treatment of GERD: a multicenter trial. Gastrointest Endosc 2001;53:416-22. [Crossref] [PubMed]
- Schiefke I, Zabel-Langhennig A, Neumann S, et al. Long term failure of endoscopic gastroplication (EndoCinch). Gut 2005;54:752-8. [Crossref] [PubMed]
- Domagk D, Menzel J, Seidel M, et al. Endoluminal gastroplasty (EndoCinch) versus endoscopic polymer implantation (Enteryx) for treatment of gastroesophageal reflux disease: 6-month results of a prospective, randomized trial. Am J Gastroenterol 2006;101:422-30. [Crossref] [PubMed]
- Abou-Rebyeh H, Hoepffner N, Rösch T, et al. Long-term failure of endoscopic suturing in the treatment of gastroesophageal reflux: a prospective follow-up study. Endoscopy 2005;37:213-6. [Crossref] [PubMed]
- Chen D, Barber C, McLoughlin P, et al. Systematic review of endoscopic treatments for gastro-oesophageal reflux disease. Br J Surg 2009;96:128-36. [Crossref] [PubMed]
- Alzahrani A, Anvari M, Dallemagne B, et al. Surgical approach after failed enteryx injection for GERD. JSLS 2007;11:97-100. [PubMed]
- Lo WK, Mashimo H. Critical Assessment of Endoscopic Techniques for Gastroesophageal Reflux Disease. J Clin Gastroenterol 2015;49:720-4. [Crossref] [PubMed]
- Fockens P, Cohen L, Edmundowicz SA, et al. Prospective randomized controlled trial of an injectable esophageal prosthesis versus a sham procedure for endoscopic treatment of gastroesophageal reflux disease. Surg Endosc 2010;24:1387-97. [Crossref] [PubMed]
- Stavropoulos SN, Modayil R, Friedel D. Current applications of endoscopic suturing. World J Gastrointest Endosc 2015;7:777-89. [Crossref] [PubMed]
- Miller C, Magarinos J, Akcelik A, et al. Endoscopic tissue approximation in clinical practice and the OverStitch device: a narrative review. Ann Esophagus 2023;6:8.
- Yanagimoto Y, Yamasaki M, Nagase H, et al. Endoscopic anti-reflux valve for post-esophagectomy reflux: an animal study. Endoscopy 2016;48:1119-24. [Crossref] [PubMed]
- Li X, Peng L, Zhang G. Successful anti-reflux treatment post-esophagectomy using endoscopic suturing with Overstitch. Dig Endosc 2019;31:e40-1. [Crossref] [PubMed]
- Satodate H, Inoue H, Yoshida T, et al. Circumferential EMR of carcinoma arising in Barrett's esophagus: case report. Gastrointest Endosc 2003;58:288-92. [Crossref] [PubMed]
- Inoue H, Ito H, Ikeda H, et al. Anti-reflux mucosectomy for gastroesophageal reflux disease in the absence of hiatus hernia: a pilot study. Ann Gastroenterol 2014;27:346-51. [PubMed]
- Yoo IK, Ko WJ, Kim HS, et al. Anti-reflux mucosectomy using a cap-assisted endoscopic mucosal resection method for refractory gastroesophageal disease: a prospective feasibility study. Surg Endosc 2020;34:1124-31. [Crossref] [PubMed]
- Debourdeau A, Vitton V, Monino L, et al. Antireflux Mucosectomy Band (ARM-b) in Treatment of Refractory Gastroesophageal Reflux Disease After Bariatric Surgery. Obes Surg 2020;30:4654-8. [Crossref] [PubMed]
- Inoue H, Tanabe M, de Santiago ER, et al. Anti-reflux mucosal ablation (ARMA) as a new treatment for gastroesophageal reflux refractory to proton pump inhibitors: a pilot study. Endosc Int Open 2020;8:E133-8. [Crossref] [PubMed]
- Inoue H, Ueno A, Shimamura Y, et al. Peroral endoscopic myotomy and fundoplication: a novel NOTES procedure. Endoscopy 2019;51:161-4. [Crossref] [PubMed]
Cite this article as: Akcelik A, Miller C, Bakhos C, Abbas A, Petrov R. Endoscopic interventions in the management of the gastroesophageal reflux: a narrative review. Ann Esophagus 2023;6:23.


